FDA schedules kids' COVID shots review
FDA advisors to review Pfizer data for 5-11 year olds on October 26, and booster shots for Moderna and J&J vaccines on the agenda for October 14 and 15. Death toll passes 700,000.
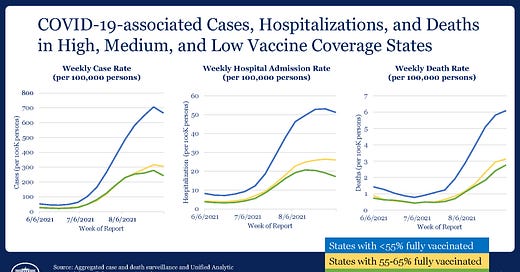
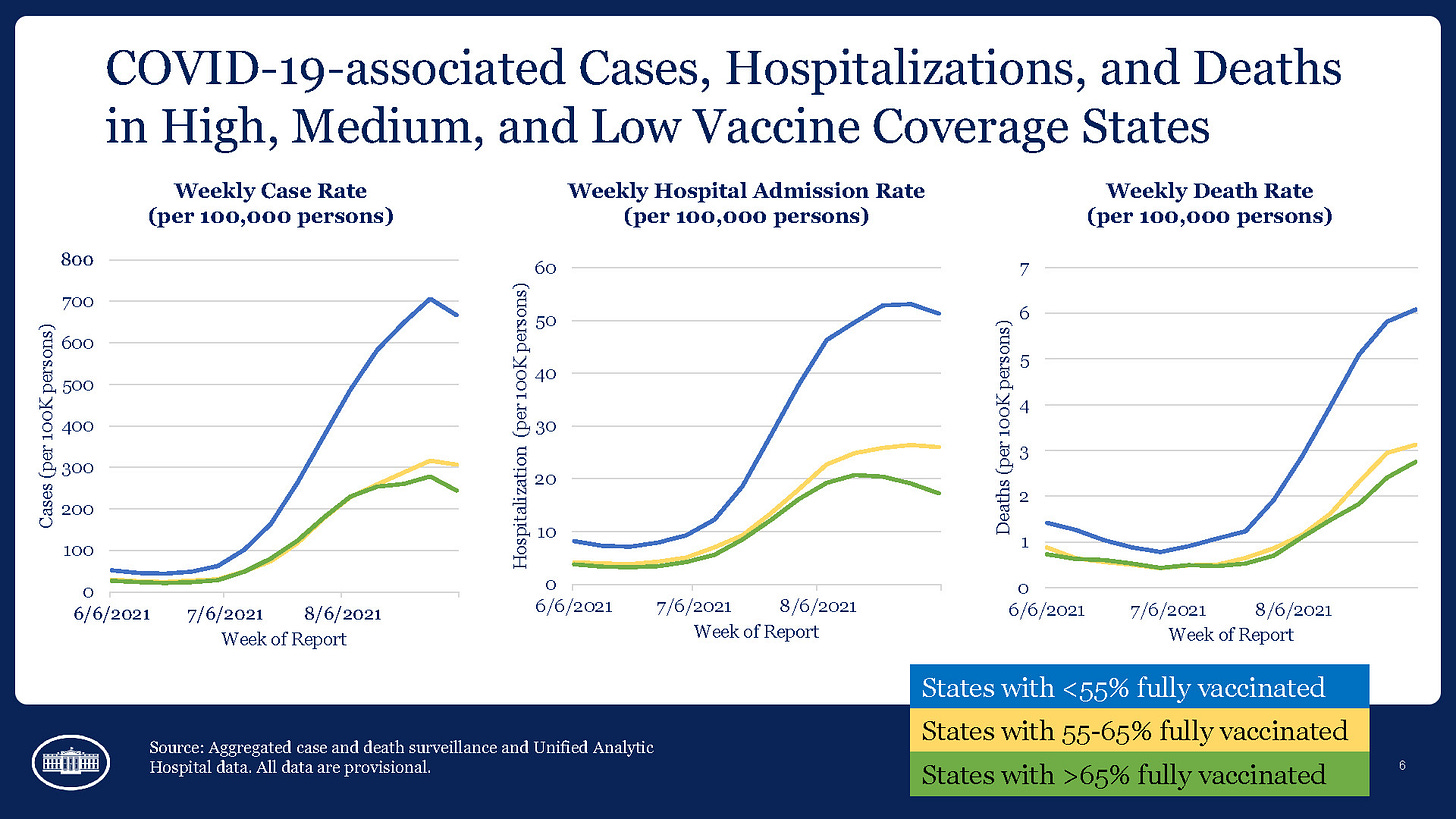
COVID-19 case and hospitalization rates are starting to trend downward, but deaths in areas with fewer than 55% of their population fully vaccinated are still climbing. CDC Director Dr. Rochelle Walensky reported that states with more than 65% fully vaccinated are seeing the most dramatic declines in new cases and hospitalizations. A study from the Seattle area estimates that unvaccinated individuals are 57 times more likely to die than those who are fully vaccinated.
The USA has lost 696,603 people to COVID-19, and the 7-day average of new cases was 106,395, which is 13% lower than a week ago, according to the CDC. Other tallies indicate the USA has already passed the 700,000 milestone.
“When you see the curve start to go down, that’s not a reason to remain unvaccinated.” - Dr. Anthony Fauci, chief White House medical advisor
The delta variant appears to be more efficient at spreading through the air than prior COVID strains, according to reporting in the New York Times.
Anti-COVID pills: Merck said it will seek emergency use authorization for molnupiravir, a pill that has been shown in a clinical trial to reduce risk of hospitalization or death from COVID-19 by 50%. The White House says the federal government will purchase 1.7 million doses for $1.2 billion, if the FDA grants the EUA.
Pfizer’s data on a dosing regimen for COVID-19 vaccinations for children 5-11 years old will be presented to and discussed by the FDA’s panel of scientific advisors on October 26, the agency announced Friday. If the advisors recommend the vaccine’s emergency use authorization for kids, the next steps are the FDA commissioner’s approval, then CDC. This likely means kids can start getting vaccinated by Halloween.
Before then, FDA and its outside experts will consider data on booster shots for both the Moderna and Johnson & Johnson COVID-19 vaccines, in meetings October 14 and 15, respectively.
Mix and match? Data from an NIH study of individuals given a Moderna COVID-19 shot as a booster 12-20 weeks after receiving any one of the three vaccines used in the USA will also be presented at the October 15 meeting.
Studies of the Janssen Ebola vaccine, which uses the same vaccine platform as the COVID-19 vaccine, have shown that boosters using a different technology may be safe and effective.
A study in Canada found that mixing the Pfizer and Moderna vaccines in frail elderly individuals did not change the resulting immune response. However, they found that antibody levels from the Moderna vaccine remained higher than those from the Pfizer vaccine between the first dose and a delayed second dose — 16 weeks after the first shot instead of 3 or 4 as currently recommended. The study has not been peer reviewed.
Natural immunity is half as good as vaccinated immunity, according to new data. Before the delta variant, the gap was even wider.


California’s governor announced vaccine requirements for kids in schools, effective the next semester after full FDA approval for each age group. This means the mandates most likely won’t take effect until Fall 2022.
Want to know your local COVID-19 risk? Here are some ways to check:
From the CDC, a new CDC COVID County Check
The New York Times publishes county-level risk estimates daily.
MicroCovid.org compiles data from multiple sources and calculates your estimated risk for specific activities.
Communicating about COVID
West Virginia health experts think they got the COVID vaccine communications right. Here’s their report:
Data-driven Communication about COVID-19 Vaccines in West Virginia | CDC
In Baltimore, the city health department has taken a different approach, and they are seeing evidence that it may be working.
ICYMI: Dr. Craig Spencer’s recent article in The Atlantic is a must-read reference for anyone who encounters someone who is anti-vaccine
And in COVID-19 politics:
Mississipi’s governor defends her diversion of COVID relief funds to build new prisons.
Airline pilots based in Texas are fighting federal vaccine requirements that apply to their jobs with American Airlines and Southwest Airlines.
Anti-mask and other anti-health efforts are being backed by some of the same people who bankroll other anti-regulatory campaigns. The Washington Post obtained a template letter circulated by a Koch family-funded organization used by parents to oppose school mask mandates. The letter makes multiple claims that are refuted by scientific studies.
That’s it for this week. Questions or comments? Please share. More next Thursday.